 
Myocardial infarction (MI) is a leading cause of death in westernized countries. These data indicate that the non-perfused area along the endocardium is a site of active angiogenesis and a promising target for MI treatment. In contrast, cardiomyocytes in the border zone’s hypoxic area underwent apoptosis within 12 h of MI, and the border area that was amenable to treatment disappeared. Moreover, enhanced angiogenesis increased cardiomyocyte survival along the endocardium in the ischemic zone and suppressed ventricular remodeling in infarcted hearts. Here, we describe a novel circulatory system wherein new vessels develop from the endocardium of the left ventricle to perfuse the hypoxic area and salvage damaged cardiomyocytes at 3–14 days after MI by activating vascular endothelial growth factor signaling. Identifying targets that benefit from angiogenesis treatment is indispensable for the development of revolutionary therapies. 
However, the mechanism and timing for new vessel formation in the mammalian heart following hypoxia are unclear. It has been speculated that the ischemic border zone is a unique area rescued by angiogenic therapy. 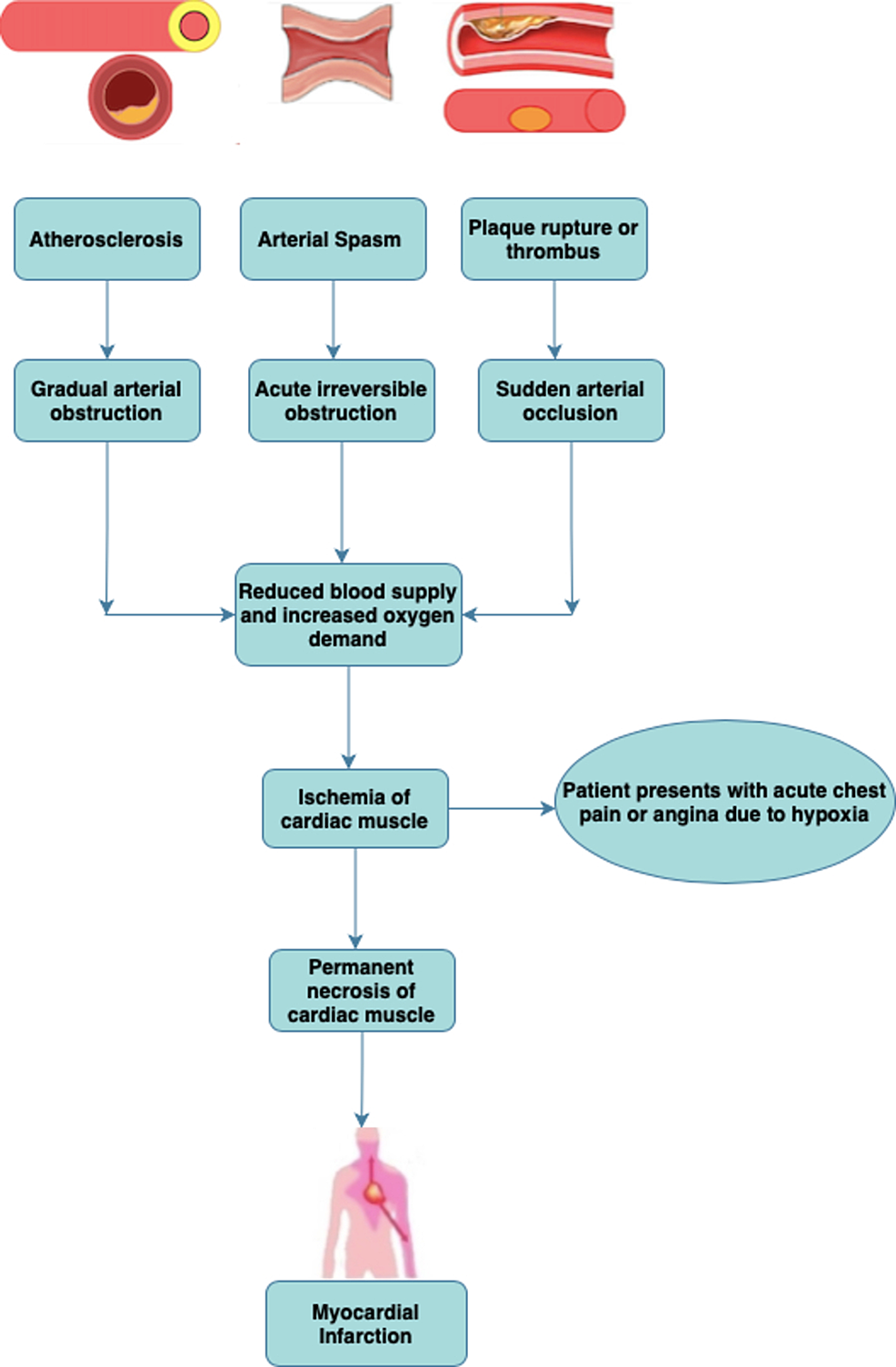
Angiogenesis can reduce MI-induced damage by mitigating hypoxia. Cardiomyocytes are susceptible to apoptosis caused by hypoxia during the acute and subacute phases of myocardial infarction (MI). 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
